Chemotherapy: Medicine or Murder?

This article on chemotherapy is a chapter from my book Cancer Cured. If you find it helpful, please support my work and buy my book on Amazon.
Most people I’ve talked to about cancer have been highly skeptical of chemotherapy as a treatment and the cancer industry in general. This is because most of us have witnessed someone in our lives undergo chemotherapy and come out looking far worse than before receiving it. Apparently the idea of using weapons and calling them treatments does not seem to sit well with most people.
When I was 12 years old my mother died of cancer. Since then I’ve made it my goal in life to ensure that no child has to go through what I did, ever again. In order to do this we all need to take an honest look at the for-profit cancer industry and the treatments it offers. The question we need to be asking ourselves is this:
Does chemotherapy (and other mainstream cancer treatments) improve the quality of life and survival of people with cancer or not?
If they do, then we should keep using them. If they don’t, and are actually making people’s health worse, then we we need to immediately ban their use in oncology centres worldwide and find a more appropriate treatment – one that facilitates healing in patients without causing any harm.
TABLE OF CONTENTS
Introduction
Chemotherapy is a cancer treatment where highly-toxic chemicals are injected into patients in an attempt to kill cancer cells. The first chemotherapeutic agent ever used, which is still being administered to this day, is a derivative of the chemical weapon mustard gas, called mustargen.
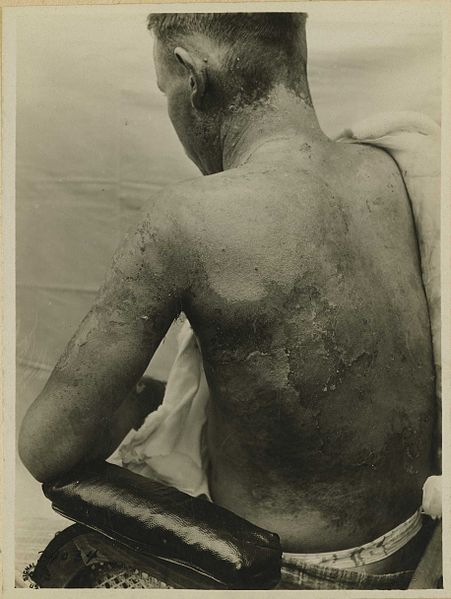
Soldier with mustard gas burns on his back and arm.
The United States learned a lot about mustard gas during World War II, where damaged bone marrow and lymph tissues seen in autopsies of exposed soldiers revealed the weapon’s prime target: the immune system.1 Even more was learned about the effects of mustard gas when the US government conducted a series of secret tests on 60,000 of its own troops. National Public Radio broke the story in 2015:
“Sixty-thousand American troops served as test subjects, and about 4,000 were used in extreme tests that government studies have linked to illnesses including skin cancer, leukemia and chronic breathing problems. The test subjects were sworn to secrecy until the program was formally declassified in 1993. By then, the youngest World War II veterans were in their 60s and 70s. Many of the men in the experiments never shared the details with their families.”2
From Battlefield to Cancer Clinic
What do you do after discovering a chemical weapon that knocks out the immune system, causes cancer and makes exposed skin literally slough off the body? Naturally you dispose of it – as safely as possible – and stop its production forever. But while mustard gas has been banned on the battlefield by international treaties,3 instead of leaving this devastating poison behind us as a dark remnant of our past to be revisited only in history books – the government decided to begin injecting it into sick people with cancer.
After World War II ended, the US Department of Defense funded Dr. Goodman and Dr. Gilman of Yale University to administer mustard gas to rats and observe its effects on tumors. Their tumors regressed. They tested it on a lymphoma patient with advanced cancer and their tumors also regressed.4-6 So amazed was the medical community that a drug could cause tumor regression, that it didn’t seem to matter the patient died within a couple of months.

The beginning of the chemotherapy testing program. J. Hartwell (right) and assistant at the National Cancer Institute, circa 1950.
Interestingly, around this same time Dr. Gerson – an American physician famous for his nutritional approach to cancer, which included fresh fruit and vegetable juices, liver extract injections, thyroid hormone, coffee enemas and other nutrients – presented cases to US congress of cancer patients he had cured using his nutritional therapy.7 The world of medicine was at a fork in the road, Dr. Nicholas Gonzales explains, “it could have gone toward natural treatments, it could have gone toward synthetic. But because of that extraordinary response in a single patient that lasted a few weeks, the entire chemo industry came into fruition.”8
Warning Label for Mustargen:
“This drug is HIGHLY TOXIC and both powder and solution must be handled and administered with care. Inhalation of dust or vapors and contact with skin or mucous membranes, especially those of the eyes, must be avoided. Avoid exposure during pregnancy. Due to the toxic properties of mechlorethamine (e.g., corrosivity, carcinogenicity, mutagenicity, teratogenicity), special handling procedures should be reviewed prior to handling and followed diligently. Extravasation of the drug into subcutaneous tissues results in a painful inflammation. The area usually becomes indurated and sloughing may occur.”18
Chemotherapy vs. Cancer
It doesn’t take more than common sense to reason that injecting poison into the veins of a sick person will 1) not cure them and 2) probably make their health worse. A study published in The Lancet in 1980 found that of 78 patients who received chemotherapy, survival “was no better than that of the 80 who did not receive chemotherapy.” Furthermore, regression of tumors was found to have no impact on survival and, “survival may even have been shortened in some patients given chemotherapy,” the study reports.9
The most comprehensive review ever conducted on the efficacy of chemotherapy was completed by German epidemiologist and biostatistician Dr. Ulrich Abel. Europe’s most popular news magazine Der Spiegel, which sells over 1-million copies per week, featured Dr. Abel’s publication in a 2004 article titled Useless Poisonous Cures (Giftkur ohne Nutzen).10 In order to obtain every study and clinical trial ever published on chemotherapy, Dr. Abel sent letters to over 350 medical centers across the world; his review consisted of thousands of studies and took two years to complete.
Dr. Abel pronounced that despite new and increasingly expensive poisons being used during chemotherapy, “patients do not live a day longer” than they did 25 years prior. Overall worldwide chemotherapy success rates he said were “appalling,” and that “for most internal cancers no proof exists that chemotherapy, especially the increasingly high dose variety, increases life expectancy or improves quality of life.” Dr. Abel estimated at least 80% of chemotherapy administered throughout the world is completely worthless.11
A group of Australian scientists published a study in 2004 suggesting that far more than just 80% of chemotherapy administered is worthless. During a follow-up with cancer patients 5-years after receiving chemotherapy, the researchers determined that only 2.1% of patients in the US and 2.3% of patients in Australia were still alive – exposing chemotherapy’s astonishing 98% failure rate.12 I wonder how many of these patients would have been alive at 5-years if they hadn’t received chemotherapy.
Seeking a greater understanding of what happens inside the body after an injection of chemotherapy, scientists from Harvard Medical School and the University of Massachusetts tested 88 currently-used chemotherapeutic drugs on fruit flies in 2013. Michelle Markstein, molecular biologist and co-author of the study reported, “…several chemotherapeutics that stop fast growing tumors have the opposite effect on stem cells in the same animal, causing them to divide too rapidly.”13 By shrinking the initial tumor mass, chemotherapy deceives doctors into thinking patients are benefitting from the treatment, when in actuality, the growth and spread of cancer are being accelerated by it.
Another way of analyzing the effects of chemotherapy on human health is to look at people who were involved in producing it during times of war. Retired Japanese poison gas factory workers were evaluated 57-years after they had been manufacturing mustard gas during World War II. The study found that exposure to mustard gas “significantly increases the long-term risk of death from respiratory cancer and chronic bronchitis/emphysema.”84
For the first time ever, researchers investigated chemotherapy-induced death at a number of hospitals in the UK. Published in the esteemed journal Lancet Oncology in 2016, the study found that 8.4% of people undergoing chemotherapy for lung cancer and 2.4% of people treated for breast cancer nationwide were killed by the treatment within 30-days of administration. When they looked at the numbers from the Milton Keynes Hospital they discovered an even more startling figure: 50.9% of lung cancer patients were killed by chemotherapy within 30-days of treatment.14
Success Stories?
One Cancer Patient’s Experience with Chemotherapy:
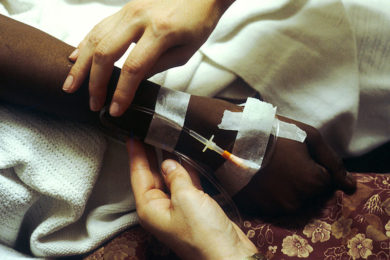 “This highly toxic fluid was being injected into my veins. The nurse administering it was wearing protective gloves because it would burn her skin if just a tiny drip came into contact with it. I couldn’t help asking myself ‘If such precautions are needed to be taken on the outside, what is it doing to me on the inside?’ From 7 pm that evening, I vomited solidly for two and a half days. During my treatment, I lost my hair by the handful, I lost my appetite, my skin colour, my zest for life. I was death on legs,” described a cancer patient in the book Now and Then by Bob Madison.
“This highly toxic fluid was being injected into my veins. The nurse administering it was wearing protective gloves because it would burn her skin if just a tiny drip came into contact with it. I couldn’t help asking myself ‘If such precautions are needed to be taken on the outside, what is it doing to me on the inside?’ From 7 pm that evening, I vomited solidly for two and a half days. During my treatment, I lost my hair by the handful, I lost my appetite, my skin colour, my zest for life. I was death on legs,” described a cancer patient in the book Now and Then by Bob Madison.
Women Struggling to Live Normal Lives Following Chemotherapy:
Thousands of women who have received chemotherapy for breast cancer are struggling to live normal lives, reports a 2016 study published in the journal Cancer.
Just one year after treatment, 20% of women above the age of 65 were so debilitated that they couldn’t carry out basic daily tasks like walking across the room, light housework, shopping, kneeling or standing long enough to shower.15
 Cancer Patient Survives Decades Until Chemotherapy:
Cancer Patient Survives Decades Until Chemotherapy:
The longest-surviving breast cancer patient of all-time was diagnosed at age 45 and lived until she was 93-years-old. Following her diagnosis, she received no treatment for 22 years, at which point doctors discovered metastatic cancer in her lungs and put her on estrogen-inhibiting drugs.
14 years later, they found cancer on her spine and put her on a different estrogen inhibitor. Years later, doctors found cancer in her liver and finally decided to try chemotherapy.
After receiving two cycles of the chemotherapy drug capecitabine, she refused further treatment due to intolerable side effects, and was dead within two years.16
Chemotherapy Additional Health Effects
Despite careful adherence to a strict set of safety protocols for handling chemotherapy drugs, including the use of personal protective equipment, more than half of nursing and pharmacy workers in a 2016 study reported complaints of dizziness simply from working with chemotherapy drugs.17
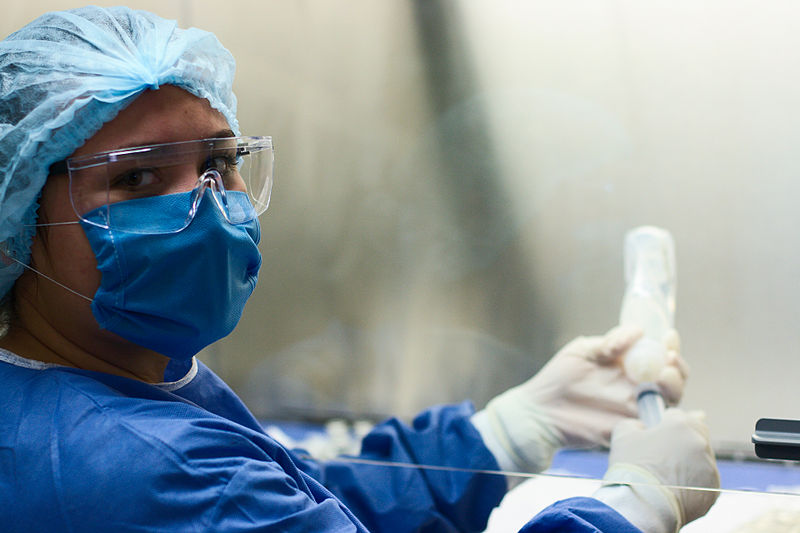
This nurse is well protected against occupational hazards of exposure to chemotherapy agents she is using a laminar flow cabinet, wearing gown, gloves, goggles and long sleeves.
Side effects of chemotherapy include, but are not limited to:
Blood Health:
- Chemotherapy decreases red blood cells (anemia)19
- Chemotherapy decreases white blood cells (leukopenia)20
- Chemotherapy decreases blood platelets (thrombocytopenia)21
Bone Health:
- Chemotherapy causes bone death (osteonecrosis)22
- Chemotherapy causes loss of bone mineral density (osteoporosis)23,24
Brain Health:
- Chemotherapy is toxic to the brain (neurotoxic)25
- Chemotherapy causes long-lasting impairment of concentration, forgetfulness and slower thinking; termed “chemobrain”26,27
- Chemotherapy causes altered consciousness28
- Chemotherapy causes degeneration of white matter in the brain (leukoencephalopathy)28
- Chemotherapy causes nerve damage (neuropathy)28
- Chemotherapy causes seizures28
- Chemotherapy causes paralysis28
- Chemotherapy causes stroke (cerebral infarction)28
Digestive Health:
- Chemotherapy causes diarrhea36
- Chemotherapy causes painful inflammation and ulceration in the digestive tract (intestinal mucositis)41
- Chemotherapy causes “significant intestinal damage in both jejunum and colon”37
Exercise:
- Chemotherapy reduces grip strength38
- Chemotherapy causes muscle dysfunction and a loss of overall strength39
Eye Health:
- Chemotherapy causes severe vision loss and altered color vision40
- Chemotherapy causes complete blindness41
Hair Health:
- Chemotherapy causes hair loss50
Healing:
- Chemotherapy impairs wound healing51
Hearing:
- Chemotherapy causes “severe to profound hearing loss”52
- Chemotherapy causes chronic ringing of the ears (tinnitus)52
Heart Health:
- Chemotherapy damages the heart53
- Chemotherapy causes heart disease54
- Chemotherapy causes heart failure55
- Chemotherapy causes heart attacks (myocardial infarction)56
Immune System:
- Chemotherapy causes long-term immune system damage57,58
- Chemotherapy exacerbates existing hepatitis C infections59
- Chemotherapy reactivates hepatitis B virus60
- Chemotherapy impairs anti-tumor immune response61
Kidney Health:
- Chemotherapy causes kidney failure65
Liver Health:
- Chemotherapy causes liver injury66
Lung Health:
- Chemotherapy causes lung disease67
Mental Health:
- Chemotherapy “decreased emotional and social function and increased distress”29
- Chemotherapy causes depression30
- Chemotherapy causes anxiety31
Oral Health:
- Chemotherapy causes severe dental caries32
- Chemotherapy causes dry mouth (xerostomia), ulcers and mouth sores68
- Chemotherapy causes oral candida (fungal) infection33
- Chemotherapy causes painful inflammation and ulceration in the mouth (oral mucositis)34
- Chemotherapy causes “a diverse spectrum of oral changes that generally are attributed to immunosuppression and bleeding tendencies”35
Pain:
- Chemotherapy causes neuropathic pain; burning or coldness, “pins and needles” sensations, numbness and itching69
- Chemotherapy pain remains one-year after treatment70
Quality of Life:
- Chemotherapy causes difficulty swallowing (dysphagia)71
- Chemotherapy causes nausea and vomiting (emesis)72,73
- Chemotherapy causes altered taste sensation74
- Chemotherapy causes migraine headaches75
Sexual Health:
- Chemotherapy causes infertility and premature ovarian failure;42,43 in up to 66% of women44
- Chemotherapy causes absence of menstrual period (amenorrhea)45
- Chemotherapy causes menopausal symptoms45
- Chemotherapy damages sperm and testicular tissue”46,47
- Chemotherapy reduces reproductive organ weight, sperm count and sperm motility46
- Chemotherapy causes “a significant decline in serum testosterone” 46
- Chemotherapy causes erectile dysfunction48,49
Skin:
- Chemotherapy causes dermatitis: itchiness, red skin, or a rash76
Sleep:
- Chemotherapy reduces sleep quality77
Thyroid Health:
- Chemotherapy “blunts thyroid function”78
- Chemotherapy impairs thyroid hormone synthesis and secretion from the thyroid gland79
- Thyroid hormones “…were remarkably altered after each cycle of chemotherapy leading to decline in thyroid function…”80
Tumor Microenvironment:
- Chemotherapy increases free radicals85
- Chemotherapy increases cortisol85
- Chemotherapy increases adrenaline93
- Chemotherapy increases prolactin94
- Chemotherapy increases estrogen64
- Chemotherapy increases tumor necrosis factor-alpha62
- Chemotherapy increases interleukin 1- beta62
- Chemotherapy increases interleukin-691
- Chemotherapy increases interleukin-892
- Chemotherapy increases nuclear factor-kappa b62
- Chemotherapy increases prostaglandins86
- Chemotherapy increases nitric oxide46
- Chemotherapy increases vascular endothelial growth factor95
- Chemotherapy increases epidermal growth factor96
- Chemotherapy increases lactic acid87
- Chemotherapy increases serotonin88
- Chemotherapy increases histamine89
- Chemotherapy increases high-mobility group box 1 protein90
Urinary Health:
- Chemotherapy causes blood in the urine (hematuria)81
- Chemotherapy causes painful urination (dysuria)81
Weight Loss:
- Chemotherapy causes muscle-wasting (cachexia)82
- “Severeloss of body weight (cachexia) is a frequent cause of death in cancer patients and is exacerbated by chemotherapy”83
Timeless Quotes
“Most cancer patients in this country die of chemotherapy. Chemotherapy does not eliminate breast, colon, or lung cancers. This fact has been documented for over a decade, yet doctors still use chemotherapy for these tumors.”
– Dr. Allen Levin, The Healing of Cancer
“As a chemist trained to interpret data, it is incomprehensible to me that physicians can ignore the clear evidence that chemotherapy does much, much more harm than good.”
– Alan Nixon, Ph.D., Past President of The American Chemical Society
“Chemotherapy and radiotherapy will make the ancient method of drilling holes in a patient’s head to permit the escape of demons look relatively advanced. Toxic chemotherapy is a hoax. The doctors who use it are guilty of pre-meditated murder, and the use of cobalt and other methods of cancer treatment popular today effectively closes the door on cure.”
– Ernst T. Krebs Jr., American Biochemist (1911-1996)
“To sell chemotherapy as a ‘therapy’ is most likely the biggest deceit in the history of medicine. Whoever masterminded this chemo-torture deserves a monument in hell.”
– Dr. Ryke Geerd Hamer, M.D.
References
- (2014). Evolution of Cancer Treatments: Chemotherapy. [Online] Available: http://www.cancer.org/cancer/cancerbasics/thehistoryofcancer/the-history-of-cancer-cancer-treatment-chemo. [November 28, 2016].
- Dickerson, C. (2015). Families React To NPR’s Reporting Of Secret Mustard Gas Testing. [Online]. Available: http://www.npr.org/2015/11/03/453962074/families-react-to-nprs-reporting-of-secret-mustard-gas-testing. [November 28, 2016].
- (2011). Chemical Weapons Specialist Helps WWII Veteran Find Effective Treatment For Mustard Gas Symptoms. [Online]. Available: http://www.cdc.gov/nceh/stories/mustard_gas.html. [November 28, 2016].
- Gilman A. Therapeutic applications of chemical warfare agents. Fed Proc. 1946;5:285-92.
Source: https://www.ncbi.nlm.nih.gov/pubmed/20990888 - Goodman LS, Wintrobe MM. Nitrogen mustard therapy; use of methyl-bis (beta-chloroethyl) amine hydrochloride and tris (beta-chloroethyl) amine hydrochloride for Hodgkin’s disease, lymphosarcoma, leukemia and certain allied and miscellaneous disorders. J Am Med Assoc. 1946;132:126-32.
Source: https://jamanetwork.com/journals/jama/article-abstract/392660 - Gilman A, Philips FS. The Biological Actions and Therapeutic Applications of the B-Chloroethyl Amines and Sulfides. Science. 1946;103(2675):409-36.
Source: http://science.sciencemag.org/content/103/2675/409 - The Gerson Institute. Gerson’s Suppressed 1946 Congressional Testimony. [Online]. Available: http://whale.to/cancer/dr_gerson.html. [November 28, 2016].
- Gonzales, N. (2012). The Shocking History of Chemotherapy. [YouTube]. Available: https://www.youtube.com/watch?v=_TptKbxQJr4. [November 28, 2016].
- Powles TJ, Coombes RC, Smith IE, Jones JM, Ford HT, Gazet JC. Failure of chemotherapy to prolong survival in a group of patients with metastatic breast cancer. 1980;1(8168 Pt 1):580-2.
Source: https://www.ncbi.nlm.nih.gov/pubmed/6102295 - Blech, Jorg. (2004). Giftkur ohne Nutzen. [Online]. Available: http://www.spiegel.de/spiegel/print/d-32362278.html [November 28, 2016].
- Abel, Ulrich, PhD “Cytostatic Therapy of Advanced Epithelial Tumors – A Critique” Lancet 10 Aug 1991.
- Morgan G, Ward R, Barton M. The contribution of cytotoxic chemotherapy to 5-year survival in adult malignancies. Clin Oncol (R Coll Radiol). 2004;16(8):549-60.
Source: https://www.sciencedirect.com/science/article/pii/S0936655504002225 - Markstein M, Dettorre S, Cho J, Neumüller RA, Craig-müller S, Perrimon N. Systematic screen of chemotherapeutics in Drosophila stem cell tumors. Proc Natl Acad Sci USA. 2014;111(12):4530-5.
Source: https://www.ncbi.nlm.nih.gov/pubmed/24616500 - Wallington M, Saxon EB, Bomb M, et al. 30-day mortality after systemic anticancer treatment for breast and lung cancer in England: a population-based, observational study. Lancet Oncol. 2016;17(9):1203-16.
Source: http://www.thelancet.com/journals/lanonc/article/PIIS1470-2045(16)30383-7/abstract - Owusu C, Margevicius S, Schluchter M, Koroukian SM, Schmitz KH, Berger NA. Vulnerable elders survey and socioeconomic status predict functional decline and death among older women with newly diagnosed nonmetastatic breast cancer. Cancer. 2016;122(16):2579-86.
Source: http://onlinelibrary.wiley.com/doi/10.1002/cncr.30046/abstract - Bayraktar S, Garcia-buitrago MT, Hurley E, Gluck S. Surviving metastatic breast cancer for 18 years: a case report and review of the literature. Breast J. 2011;17(5):521-4.
Source: https://www.ncbi.nlm.nih.gov/pubmed/21726350 - Fernandes NM, Pelissari IG, Cogo LA, Santos filha VA. Workplace Activity in Health Professionals Exposed to Chemotherapy Drugs: An Otoneurological Perspective. Int Arch Otorhinolaryngol. 2016;20(4):331-338.
Source: https://www.researchgate.net/publication/294276617_Workplace_Activity_in_Health_Professionals_Exposed_to_Chemotherapy_Drugs_An_Otoneurological_Perspective - Drugs.com. (2013). Mustargen. [Online]. Available: https://www.drugs.com/pro/mustargen.html [November 28, 2016].
- Mountzios G, Aravantinos G, Alexopoulou Z, et al. Lessons from the past: Long-term safety and survival outcomes of a prematurely terminated randomized controlled trial on prophylactic vs. hemoglobin-based administration of erythropoiesis-stimulating agents in patients with chemotherapy-induced anemia. Mol Clin Oncol. 2016;4(2):211-220.
Source: https://spandidos-publications.com/mco/4/2/211 - Li DY, Yu TT, Bai H, Chen XY. [Clinical study on effect of compound granule prescription of thunberg fritillary bulb in relieving post-chemotherapy bone marrow suppression in RAL patients]. Zhongguo Zhong Yao Za Zhi. 2012;37(20):3155-7.
Source: https://www.ncbi.nlm.nih.gov/pubmed/23311172 - Tamamyan G, Danielyan S, Lambert MP. Chemotherapy induced thrombocytopenia in pediatric oncology. Crit Rev Oncol Hematol. 2016;99:299-307.
Source: http://www.sciencedirect.com/science/article/pii/S1040842816300051 - Antonuzzo L, Lunghi A, Giommoni E, Brugia M, Di costanzo F. Regorafenib Also Can Cause Osteonecrosis of the Jaw. J Natl Cancer Inst. 2016;108(4).
Source: https://academic.oup.com/jnci/article/108/4/djw002/2412605 - Bordbar MR, Haghpanah S, Dabbaghmanesh MH, Omrani GR, Saki F. Bone mineral density in children with acute leukemia and its associated factors in Iran: a case-control study. Arch Osteoporos. 2016;11(1):36.
Source: https://www.ncbi.nlm.nih.gov/pubmed/27785744 - Lee SW, Yeo SG, Oh IH, Yeo JH, Park DC. Bone mineral density in women treated for various types of gynecological cancer. Asia Pac J Clin Oncol. 2016.
Source: https://www.researchgate.net/publication/306090692_Bone_mineral_density_in_women_treated_for_various_types_of_gynecological_cancer - Matsuoka A, Mitsuma A, Maeda O, et al. Quantitative assessment of chemotherapy-induced peripheral neurotoxicity using a point-of-care nerve conduction device. Cancer Sci. 2016;107(10):1453-1457.
Source: http://onlinelibrary.wiley.com/doi/10.1111/cas.13010/abstract - Iarkov A, Appunn D, Echeverria V. Post-treatment with cotinine improved memory and decreased depressive-like behavior after chemotherapy in rats. Cancer Chemother Pharmacol. 2016;78(5):1033-1039.
Source: https://link.springer.com/article/10.1007/s00280-016-3161-0 - Rendeiro C, Sheriff A, Bhattacharya TK, et al. Long-lasting impairments in adult neurogenesis, spatial learning and memory from a standard chemotherapy regimen used to treat breast cancer. Behav Brain Res. 2016;315:10-22.
Source: https://www.sciencedirect.com/science/article/pii/S0166432816304788 - Reddy AT, Witek K. Neurologic complications of chemotherapy for children with cancer. Curr Neurol Neurosci Rep. 2003;3(2):137-42.
Source: https://www.ncbi.nlm.nih.gov/pubmed/12583842 - Dunn J, Watson M, Aitken JF, Hyde MK. Systematic Review of Psychosocial Outcomes for Patients with Advanced Melanoma. Psychooncology. 2016.
Source: http://onlinelibrary.wiley.com/doi/10.1002/pon.4290/abstract - Oh PJ, Lee JR. [Effect of Cancer Symptoms and Fatigue on Chemotherapy-related Cognitive Impairment and Depression in People with Gastrointestinal Cancer]. J Korean Acad Nurs. 2016;46(3):420-30.
Source: https://www.researchgate.net/publication/305310719_Effect_of_Cancer_Symptoms_and_Fatigue_on_Chemotherapy-related_Cognitive_Impairment_and_Depression_in_People_with_Gastrointestinal_Cancer - Baek Y, Yi M. [Factors Influencing Quality of Life during Chemotherapy for Colorectal Cancer Patients in South Korea]. J Korean Acad Nurs. 2015;45(4):604-12.
Source: http://europepmc.org/abstract/MED/26364535 - Peretz B, Sarnat H, Kharouba J. Chemotherapy induced dental changes in a child with medulloblastoma: a case report. J Clin Pediatr Dent. 2014;38(3):251-4.
Source: https://www.ncbi.nlm.nih.gov/pubmed/25095321 - Alt-epping B, Nejad RK, Jung K, Gross U, Nauck F. Symptoms of the oral cavity and their association with local microbiological and clinical findings–a prospective survey in palliative care. Support Care Cancer. 2012;20(3):531-7.
Source: https://link.springer.com/article/10.1007/s00520-011-1114-z - Dreizen S, Mccredie KB, Keating MJ. Chemotherapy-induced oral mucositis in adult leukemia. Postgrad Med. 1981;69(2):103-8, 111-2.
Source: https://www.ncbi.nlm.nih.gov/pubmed/7454641 - Farsi DJ. Children Undergoing Chemotherapy: Is It Too Late for Dental Rehabilitation?. J Clin Pediatr Dent. 2016;40(6):503-505.
Source: https://www.ncbi.nlm.nih.gov/pubmed/27805886 - Abalo R, Uranga JA, Pérez-garcía I, et al. May cannabinoids prevent the development of chemotherapy-induced diarrhea and intestinal mucositis? Experimental study in the rat. Neurogastroenterol Motil. 2016.
Source: http://onlinelibrary.wiley.com/doi/10.1111/nmo.12952/full - Forsgård RA, Korpela R, Holma R, et al. Intestinal permeability to iohexol as an in vivo marker of chemotherapy-induced gastrointestinal toxicity in Sprague-Dawley rats. Cancer Chemother Pharmacol. 2016;78(4):863-74.
Source: https://link.springer.com/article/10.1007/s00280-016-3150-3 - Klepin HD, Tooze JA, Pardee TS, et al. Effect of Intensive Chemotherapy on Physical, Cognitive, and Emotional Health of Older Adults with Acute Myeloid Leukemia. J Am Geriatr Soc. 2016;64(10):1988-1995.
Source: http://onlinelibrary.wiley.com/doi/10.1111/jgs.14301/full - Gilliam LA, Lark DS, Reese LR, et al. Targeted overexpression of mitochondrial catalase protects against cancer chemotherapy-induced skeletal muscle dysfunction. Am J Physiol Endocrinol Metab. 2016;311(2):E293-301.
Source: https://www.medscape.com/medline/abstract/27329802 - Giralt J, Rey A, Villanueva R, Alforja S, Casaroli-marano RP. Severe visual loss in a breast cancer patient on chemotherapy. Med Oncol. 2012;29(4):2567-9.
Source: https://link.springer.com/article/10.1007/s12032-012-0191-2 - Cloutier AO. Ocular side effects of chemotherapy: nursing management. Oncol Nurs Forum. 1992;19(8):1251-9.
Source: https://www.ncbi.nlm.nih.gov/pubmed/1408967 - Batchvarov IS, Taylor RW, Bustamante-marín X, et al. A grafted ovarian fragment rescues host fertility after chemotherapy. Mol Hum Reprod. 2016.
Source: https://www.researchgate.net/publication/308834484_A_grafted_ovarian_fragment_rescues_host_fertility_after_chemotherapy - Clowse ME, Behera MA, Anders CK, et al. Ovarian preservation by GnRH agonists during chemotherapy: a meta-analysis. J Womens Health (Larchmt). 2009;18(3):311-9.
Source: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2858300/ - Garrido-oyarzún MF, Castelo-branco C. Controversies over the use of GnRH agonists for reduction of chemotherapy-induced gonadotoxicity. Climacteric. 2016;19(6):522-525.
Source: https://www.researchgate.net/publication/308280085_Controversies_over_the_use_of_GnRH_agonists_for_reduction_of_chemotherapy-induced_gonadotoxicity - Li CY, Chen ML. [Chemotherapy-Induced Amenorrhea and Menopause Symptoms in Women With Breast Cancer]. Hu Li Za Zhi. 2016;63(5):19-26.
Source: https://www.researchgate.net/publication/309257609_Chemotherapy-Induced_Amenorrhea_and_Menopause_Symptoms_in_Women_With_Breast_Cancer - Eid AH, Abdelkader NF, Abd el-raouf OM, Fawzy HM, El-denshary ES. Carvedilol alleviates testicular and spermatological damage induced by cisplatin in rats via modulation of oxidative stress and inflammation. Arch Pharm Res. 2016.
Source: https://link.springer.com/article/10.1007/s12272-016-0833-6 - Levi M, Popovtzer A, Tzabari M, et al. Cetuximab intensifies cisplatin-induced testicular toxicity. Reprod Biomed Online. 2016;33(1):102-10.
Source: https://www.sciencedirect.com/science/article/pii/S1472648316300670 - Huang M, Lin J, Yu X, et al. Erectile and urinary function in men with rectal cancer treated by neoadjuvant chemoradiotherapy and neoadjuvant chemotherapy alone: a randomized trial report. Int J Colorectal Dis. 2016;31(7):1349-57.
Source: https://link.springer.com/article/10.1007/s00384-016-2605-7 - Annam K, Voznesensky M, Kreder KJ. Understanding and Managing Erectile Dysfunction in Patients Treated for Cancer. J Oncol Pract. 2016;12(4):297-304.
Source: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5015452/ - Trüeb RM. Chemotherapy-induced alopecia. Semin Cutan Med Surg. 2009;28(1):11-4.
Source: https://www.sciencedirect.com/science/article/pii/S0190962211003124 - Tanaka S, Hayek G, Jayapratap P, et al. The Impact of Chemotherapy on Complications Associated with Mastectomy and Immediate Autologous Tissue Reconstruction. Am Surg. 2016;82(8):713-7.
Source: https://www.medscape.com/medline/abstract/27657587 - Frisina RD, Wheeler HE, Fossa SD, et al. Comprehensive Audiometric Analysis of Hearing Impairment and Tinnitus After Cisplatin-Based Chemotherapy in Survivors of Adult-Onset Cancer. J Clin Oncol. 2016;34(23):2712-20.
Source: http://ascopubs.org/doi/full/10.1200/JCO.2016.66.8822 - Truong J, Yan AT, Cramarossa G, Chan KK. Chemotherapy-induced cardiotoxicity: detection, prevention, and management. Can J Cardiol. 2014;30(8):869-78.
Source: https://www.sciencedirect.com/science/article/pii/S0828282X14003158 - Granger CB. Prediction and prevention of chemotherapy-induced cardiomyopathy: can it be done?. Circulation. 2006;114(23):2432-3.
Source: http://circ.ahajournals.org/content/114/23/2432.long - Smith SA, Auseon AJ. Chemotherapy-induced takotsubo cardiomyopathy. Heart Fail Clin. 2013;9(2):233-42.
Source: http://www.heartfailure.theclinics.com/article/S1551-7136(12)00122-5/fulltext - Sakka S, Kawai K, Tsujimoto I, et al. [Severe Acute Myocardial Infarction during Induction Chemotherapy for Retroperitoneal Germ Cell Tumor : A Case Report]. Hinyokika Kiyo. 2016;62(9):483-487.
Source: https://www.researchgate.net/publication/316698854_Severe_Acute_Myocardial_Infarction_during_Induction_Chemotherapy_for_Retroperitoneal_Germ_Cell_Tumor_A_Case_Report - Perkins JL, Harris A, Pozos TC. Immune Dysfunction After Completion of Childhood Leukemia Therapy. J Pediatr Hematol Oncol. 2016.
Source: http://journals.lww.com/jpho-online/Abstract/2017/01000/Immune_Dysfunction_After_Completion_of_Childhood.1.aspx - Verma R, Foster RE, Horgan K, et al. Lymphocyte depletion and repopulation after chemotherapy for primary breast cancer. Breast Cancer Res. 2016;18(1):10.
Source: https://www.ncbi.nlm.nih.gov/pubmed/26810608 - Lin JW, Chang ML, Hsu CW, et al. Acute exacerbation of hepatitis C in hepatocellular carcinoma patients receiving chemotherapy. J Med Virol. 2017;89(1):153-160.
Source: http://onlinelibrary.wiley.com/doi/10.1002/jmv.24595/abstract - Qin L, Wang F, Zou BW, Ding ZY. Chemotherapy-induced fatal hepatitis B virus reactivation in a small-cell lung cancer patient. Mol Clin Oncol. 2016;5(4):382-384.
Source: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5038378/ - Wu Y, Deng Z, Wang H, Ma W, Zhou C, Zhang S. Repeated cycles of 5-fluorouracil chemotherapy impaired anti-tumor functions of cytotoxic T cells in a CT26 tumor-bearing mouse model. BMC Immunol. 2016;17(1):29.
Source: https://www.ncbi.nlm.nih.gov/pubmed/27645787 - Li W, Yan MH, Liu Y, et al. Ginsenoside Rg5 Ameliorates Cisplatin-Induced Nephrotoxicity in Mice through Inhibition of Inflammation, Oxidative Stress, and Apoptosis. Nutrients. 2016;8(9).
Source: https://www.ncbi.nlm.nih.gov/pubmed/27649238 - El-naga RN. Pre-treatment with cardamonin protects against cisplatin-induced nephrotoxicity in rats: impact on NOX-1, inflammation and apoptosis. Toxicol Appl Pharmacol. 2014;274(1):87-95.
Source: https://www.sciencedirect.com/science/article/pii/S0041008X13004791 - Tiong V, Rozita AM, Taib NA, Yip CH, Ng CH. Incidence of chemotherapy-induced ovarian failure in premenopausal women undergoing chemotherapy for breast cancer. World J Surg. 2014;38(9):2288-96.
Source: https://link.springer.com/article/10.1007/s00268-014-2542-y - Dugbartey GJ, Peppone LJ, De graaf IA. An integrative view of cisplatin-induced renal and cardiac toxicities: Molecular mechanisms, current treatment challenges and potential protective measures. Toxicology. 2016;371:58-66.
Source: https://www.sciencedirect.com/science/article/pii/S0300483X16302360 - Hiroshima Y, Shuto K, Yamazaki K, et al. Fractal Dimension of Tc-99m DTPA GSA Estimates Pathologic Liver Injury due to Chemotherapy in Liver Cancer Patients. Ann Surg Oncol. 2016;23(13):4384-4391.
Source: https://link.springer.com/article/10.1245/s10434-016-5441-7 - Limper AH. Chemotherapy-induced lung disease. Clin Chest Med. 2004;25(1):53-64.
Source: http://williams.medicine.wisc.edu/chemolungtox.pdf - Berger velten D, Zandonade E, Monteiro de barros miotto MH. Prevalence of oral manifestations in children and adolescents with cancer submitted to chemotherapy. BMC Oral Health. 2016;16(1):107.
Source: https://link.springer.com/article/10.1186/s12903-016-0300-2 - Gong SS, Li YX, Zhang MT, et al. Neuroprotective Effect of Matrine in Mouse Model of Vincristine-Induced Neuropathic Pain. Neurochem Res. 2016;41(11):3147-3159.
Source: https://link.springer.com/article/10.1007/s11064-016-2040-8 - Hellerstedt-börjesson S, Nordin K, Fjällskog ML, Holmström IK, Arving C. Women Treated for Breast Cancer Experiences of Chemotherapy-Induced Pain: Memories, Any Present Pain, and Future Reflections. Cancer Nurs. 2016;39(6):464-472.
Source: https://www.medscape.com/medline/abstract/26632880 - Mcnamara MJ, Adelstein DJ, Allende DS, et al. Persistent Dysphagia After Induction Chemotherapy in Patients with Esophageal Adenocarcinoma Predicts Poor Post-Operative Outcomes. J Gastrointest Cancer. 2016.
Source: https://link.springer.com/article/10.1007/s12029-016-9881-x - Fakhfouri G, Mousavizadeh K, Mehr SE, et al. From Chemotherapy-Induced Emesis to Neuroprotection: Therapeutic Opportunities for 5-HT3 Receptor Antagonists. Mol Neurobiol. 2015;52(3):1670-9.
Source: https://www.ncbi.nlm.nih.gov/pubmed/25377794 - Thamlikitkul L, Srimuninnimit V, Akewanlop C, et al. Efficacy of ginger for prophylaxis of chemotherapy-induced nausea and vomiting in breast cancer patients receiving adriamycin-cyclophosphamide regimen: a randomized, double-blind, placebo-controlled, crossover study. Support Care Cancer. 2016.
Source: https://link.springer.com/article/10.1007/s00520-016-3423-8 - Sozeri E, Kutluturkan S. The Validity and Reliability of Turkish Version of the Chemotherapy-induced Taste Alteration Scale (CiTAS). Clin Nurs Res. 2016.
Source: http://journals.sagepub.com/doi/abs/10.1177/1054773816662436 - Rajasekhar A, George TJ. Gemcitabine-induced reversible posterior leukoencephalopathy syndrome: a case report and review of the literature. Oncologist. 2007;12(11):1332-5.
Source: http://theoncologist.alphamedpress.org/content/12/11/1332.full - Esquirol caussa J, Herrero vila E. [Epidermal growth factor, innovation and safety]. Med Clin (Barc). 2015;145(7):305-12.
Source: https://www.sciencedirect.com/science/article/pii/S2387020616000930 - Bagheri-nesami M, Goudarzian AH, Jan babaei G, Badiee M, Mousavi M, Sadegh sharifi M. Sleep Quality and Associated Risk Factors in Leukemia Patients Undergoing Chemotherapy in Iran. Asian Pac J Cancer Prev. 2016;17 Spec No.:107-11.
Source: https://www.researchgate.net/publication/311571038_Sleep_Quality_and_Associated_Risk_Factors_in_Leukemia_Patients_Undergoing_Chemotherapy_in_Iran - De groot S, Janssen LG, Charehbili A, et al. Thyroid function alters during neoadjuvant chemotherapy in breast cancer patients: results from the NEOZOTAC trial (BOOG 2010-01). Breast Cancer Res Treat. 2015;149(2):461-6.
Source: https://link.springer.com/article/10.1007/s10549-014-3256-4 - Anker GB, Lønning PE, Aakvaag A, Lien EA. Thyroid function in postmenopausal breast cancer patients treated with tamoxifen. Scand J Clin Lab Invest. 1998;58(2):103-7.
Source: https://www.ncbi.nlm.nih.gov/pubmed/9587161 - Khan MA, Bhurani D, Agarwal NB. Alteration of Thyroid Function in Indian HER 2-Negative Breast Cancer Patients Undergoing Chemotherapy. Asian Pac J Cancer Prev. 2015;16(17):7701-5.
Source: https://www.researchgate.net/publication/285586152_Alteration_of_Thyroid_Function_in_Indian_HER_2-Negative_Breast_Cancer_Patients_Undergoing_Chemotherapy - Degener S, Pohle A, Strelow H, et al. Long-term experience of hyperbaric oxygen therapy for refractory radio- or chemotherapy-induced haemorrhagic cystitis. BMC Urol. 2015;15:38.
Source: https://link.springer.com/article/10.1186/s12894-015-0035-4 - Nissinen TA, Degerman J, Räsänen M, et al. Systemic blockade of ACVR2B ligands prevents chemotherapy-induced muscle wasting by restoring muscle protein synthesis without affecting oxidative capacity or atrogenes. Sci Rep. 2016;6:32695.
Source: http://adsabs.harvard.edu/abs/2016NatSR…632695N - Klose R, Krzywinska E, Castells M, et al. Targeting VEGF-A in myeloid cells enhances natural killer cell responses to chemotherapy and ameliorates cachexia. Nat Commun. 2016;7:12528.
Source: https://www.nature.com/articles/ncomms12528 - Mukaida K, Hattori N, Iwamoto H, et al. Mustard gas exposure and mortality among retired workers at a poisonous gas factory in Japan: a 57-year follow-up cohort study. Occup Environ Med. 2016.
Source: http://oem.bmj.com/content/74/5/321 - Limberaki E, Eleftheriou P, Gasparis G, Karalekos E, Kostoglou V, Petrou C. Cortisol levels and serum antioxidant status following chemotherapy. 2012; Available at: https://www.researchgate.net/publication/224943223_Cortisol_levels_and_serum_antioxidant_status_following_chemotherapy. [December 1, 2016].
Source: http://www.scirp.org/journal/PaperInformation.aspx?paperID=6766 - Lalla RV, Pilbeam CC, Walsh SJ, Sonis ST, Keefe DM, Peterson DE. Role of the cyclooxygenase pathway in chemotherapy-induced oral mucositis: a pilot study. Support Care Cancer. 2010;18(1):95-103.
Source: https://link.springer.com/article/10.1007/s00520-009-0635-1 - Kondo K, Fujiwara M, Murase M, et al. Severe acute metabolic acidosis and Wernicke’s encephalopathy following chemotherapy with 5-fluorouracil and cisplatin: case report and review of the literature. Jpn J Clin Oncol. 1996;26(4):234-6.
Source: https://www.ncbi.nlm.nih.gov/pubmed/8765181 - Cubeddu LX, Hoffmann IS, Fuenmayor NT, Malave JJ. Changes in serotonin metabolism in cancer patients: its relationship to nausea and vomiting induced by chemotherapeutic drugs. Br J Cancer. 1992;66(1):198-203.
Source: https://www.ncbi.nlm.nih.gov/pubmed/1379060 - Eschalier A, Lavarenne J, Burtin C, Renoux M, Chapuy E, Rodriguez M. Study of histamine release induced by acute administration of antitumor agents in dogs. Cancer Chemother Pharmacol. 1988;21(3):246-50.
Source: https://www.researchgate.net/publication/226870597_Study_of_histamine_induced_by_acute_administration_of_antitumor_agents_in_dogs - Nguyen A, Bhavsar S, Riley E, Caponetti G, Agrawal D. Clinical Value of High Mobility Group Box 1 and the Receptor for Advanced Glycation End-products in Head and Neck Cancer: A Systematic Review. Int Arch Otorhinolaryngol. 2016;20(4):382-389.
Source: https://www.thieme-connect.com/products/ejournals/pdf/10.1055/s-0036-1583168.pdf - Cheung YT, Ng T, Shwe M, et al. Association of proinflammatory cytokines and chemotherapy-associated cognitive impairment in breast cancer patients: a multi-centered, prospective, cohort study. Ann Oncol. 2015;26(7):1446-51.
Source: https://www.ncbi.nlm.nih.gov/pubmed/25922060 - Waugh DJ, Wilson C. The interleukin-8 pathway in cancer. Clin Cancer Res. 2008;14(21):6735-41.
Source: http://clincancerres.aacrjournals.org/content/14/21/6735 - Tiligada E. Chemotherapy: induction of stress responses. Endocr Relat Cancer. 2006;13 Suppl 1(Supplement 1):S115-24.
Source: http://erc.endocrinology-journals.org/content/13/Supplement_1/S115.full - Hinterberger-fischer M, Ogris E, Kier P, et al. Elevation of plasma prolactin in patients undergoing autologous blood stem-cell transplantation for breast cancer: is its modulation a step toward posttransplant immunotherapy?. Am J Clin Oncol. 2000;23(4):325-9.
Source: https://www.researchgate.net/publication/271732855_Elevation_of_Plasma_Prolactin_in_Patients_Undergoing_Autologous_Blood_Stem-Cell_Transplantation_for_Breast_Cancer - Fan F, Gray MJ, Dallas NA, et al. Effect of chemotherapeutic stress on induction of vascular endothelial growth factor family members and receptors in human colorectal cancer cells. Mol Cancer Ther. 2008;7(9):3064-70.
Source: http://mct.aacrjournals.org/content/7/9/3064 - Wang F, Liu R, Lee SW, Sloss CM, Couget J, Cusack JC. Heparin-binding EGF-like growth factor is an early response gene to chemotherapy and contributes to chemotherapy resistance. Oncogene. 2007;26(14):2006-16.
Source: https://www.nature.com/articles/1209999
